 So, it's probably the anhydrous version dissolved in water, and the amount of heat produced might explain why iodine is a gaseous product, and not a solid. Corundum is the most common naturally occurring crystalline form of. Corundum (-aluminum oxide), emery, sapphire, amethyst, topaz, as well as many other names are reflecting its widespread occurrence in nature and industry. We should note, though, that aluminum iodide is violently reactive in water unless it's a hexahydrate. Aluminum oxide, with the chemical formula Al2O3 A l 2 O 3, is an amphoteric oxide and is commonly referred to as alumina. 
Thus, I have 6 #"I"# on the left, and I had to triple #"I"_2# on the right. Naturally, now we have two #"Al"# on the right, so I doubled the #"AlI"_3# on the left. Knowing that there were two chlorines on the left, I just found the common multiple of 2 and 3 to be 6, and doubled the #"AlCl"_3# on the right. Although I would expect iodine to be a solid. 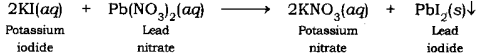
You get #"AlI"_3#.įor similar reasons, aluminum chloride is #"AlCl"_3#.Ĭhlorine and iodine both exist naturally (in their elemental states) as diatomic elements, so they are #"Cl"_2(g)# and #"I"_2(g)#, respectively. 
Al2O3 + H2SO4 Al2 (SO4)3 + H2O is a Double Displacement (Metathesis) reaction where one mole of Aluminum Oxide Al 2 O 3 and three moles of Sulfuric Acid H 2 SO 4 react to form one mole of Aluminum Sulfate Al 2 (SO 4) 3 and three moles of Water H 2 O Show. So, three iodides can bond with one aluminum. Aluminum Oxide + Sulfuric Acid Aluminum Sulfate + Water. Aluminum has a common oxidation state of #"3+"#, and that of iodine is #"1-"#. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
